How GLP-1 Receptor Agonists Work: A Research Overview
GLP-1 receptor agonists are a class of synthetic peptides that mimic glucagon-like peptide-1, a naturally occurring hormone produced in the gut. In research contexts, these compounds bind to GLP-1 receptors throughout the body, triggering a cascade of metabolic effects that include insulin stimulation, appetite suppression, and slowed gastric emptying. Understanding how GLP-1 works at the molecular level is foundational to current metabolic pathway research.
What Are GLP-1 Receptor Agonists?
Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by L-cells in the small intestine in response to food intake. In its endogenous form, it has a half-life of only one to two minutes, rapidly degraded by the enzyme dipeptidyl peptidase-4 (DPP-4). Synthetic receptor agonists were developed to replicate GLP-1’s biological activity while resisting that rapid enzymatic breakdown.
GLP-1 receptor agonists are peptide compounds engineered to activate the GLP-1 receptor (GLP-1R), a G protein-coupled receptor expressed across multiple tissue types. Activation of this receptor produces downstream signaling effects relevant to glucose homeostasis, energy balance, and body weight regulation, all of which are active areas of scientific investigation.
Compounds in this class include semaglutide, liraglutide, exenatide, and more recently tirzepatide, which adds GIP receptor agonism to GLP-1R activation. Retatrutide extends this further, targeting three receptor pathways simultaneously. Researchers studying metabolic disorders, obesity, and type 2 diabetes-related pathophysiology rely on these research compounds to probe those mechanisms in laboratory settings.
The Biological Mechanism: How GLP-1 Works Step by Step
Understanding the mechanism requires following the signaling pathway from receptor binding through downstream metabolic effects. Below is a breakdown of the four primary pathways activated by GLP-1 receptor agonists.
1. The Incretin Pathway and cAMP Signaling
When a GLP-1 receptor agonist binds to GLP-1R, it activates adenylate cyclase through a Gs protein-coupled mechanism. This produces cyclic adenosine monophosphate (cAMP), which serves as the primary intracellular second messenger driving the compound’s metabolic effects. Elevated cAMP activates protein kinase A (PKA) and exchange protein activated by cAMP (Epac2), both of which are central to downstream insulin signaling.
This cAMP-dependent pathway is dose-responsive and glucose-dependent in pancreatic beta cells, a key feature that distinguishes GLP-1 receptor agonists from other insulin-modulating research compounds. The glucose dependency of the mechanism is a subject of ongoing research interest, particularly in understanding hypoglycemia risk profiles.
2. Insulin Secretion Modulation
In pancreatic beta cells, GLP-1R activation via the cAMP/PKA pathway potentiates glucose-stimulated insulin secretion (GSIS). The mechanism works as follows: elevated cAMP enhances the closure of ATP-sensitive potassium (K-ATP) channels, causing membrane depolarization. This triggers voltage-gated calcium channel opening, intracellular calcium influx, and exocytosis of insulin-containing granules.
Critically, this effect is glucose-dependent. In low-glucose conditions, GLP-1R activation does not trigger significant insulin release, because the K-ATP channels remain open in the absence of ATP from glucose oxidation. This glucose-gating of the mechanism has made GLP-1 receptor agonists a subject of intense research interest for studies of beta cell function, glucose-stimulated insulin kinetics, and pancreatic physiology.
Research published by Drucker and Nauck in the journal Diabetologia (2006) identified this glucose-dependent mechanism as central to GLP-1’s profile and established the framework that has guided receptor agonist research since.
3. Gastric Emptying and Gastrointestinal Signaling
GLP-1 receptors are expressed in the enteric nervous system and on vagal afferent neurons innervating the gastrointestinal tract. Activation of peripheral GLP-1Rs through these pathways reduces gastric motility and slows the rate of gastric emptying. From a research standpoint, this delays nutrient absorption kinetics, which in turn blunts postprandial blood sugar level excursions.
Slowed gastric emptying is mediated in part through vagal nerve signaling rather than direct gastrointestinal smooth muscle effects, a distinction that has driven research into central nervous system-peripheral axis interactions. Studies examining GLP-1R expression in enteric neurons continue to map the precise anatomical pathways involved.
4. Appetite Signaling in the Central Nervous System
GLP-1 receptors are expressed in multiple brain regions, including the hypothalamus, brainstem (particularly the nucleus tractus solitarius), and the limbic system. When GLP-1 receptor agonists reach central GLP-1Rs, they suppress appetite through several converging mechanisms.
In the hypothalamus, GLP-1R activation reduces activity in orexigenic (appetite-stimulating) neuropeptide Y (NPY) and agouti-related peptide (AgRP) neurons while increasing activity in anorexigenic neurons. This shifts the hypothalamic energy-balance circuit toward reduced food-seeking behavior and caloric intake in preclinical models.
Research by Secher and colleagues published in Nature Medicine (2014) demonstrated that GLP-1R agonists act directly on hypothalamic neurons in rodent models, providing a mechanistic explanation for the appetite reduction observed in metabolic research. This CNS-mediated appetite suppression pathway is now considered a primary research target in obesity-related studies.
Key GLP-1 Compounds Used in Research
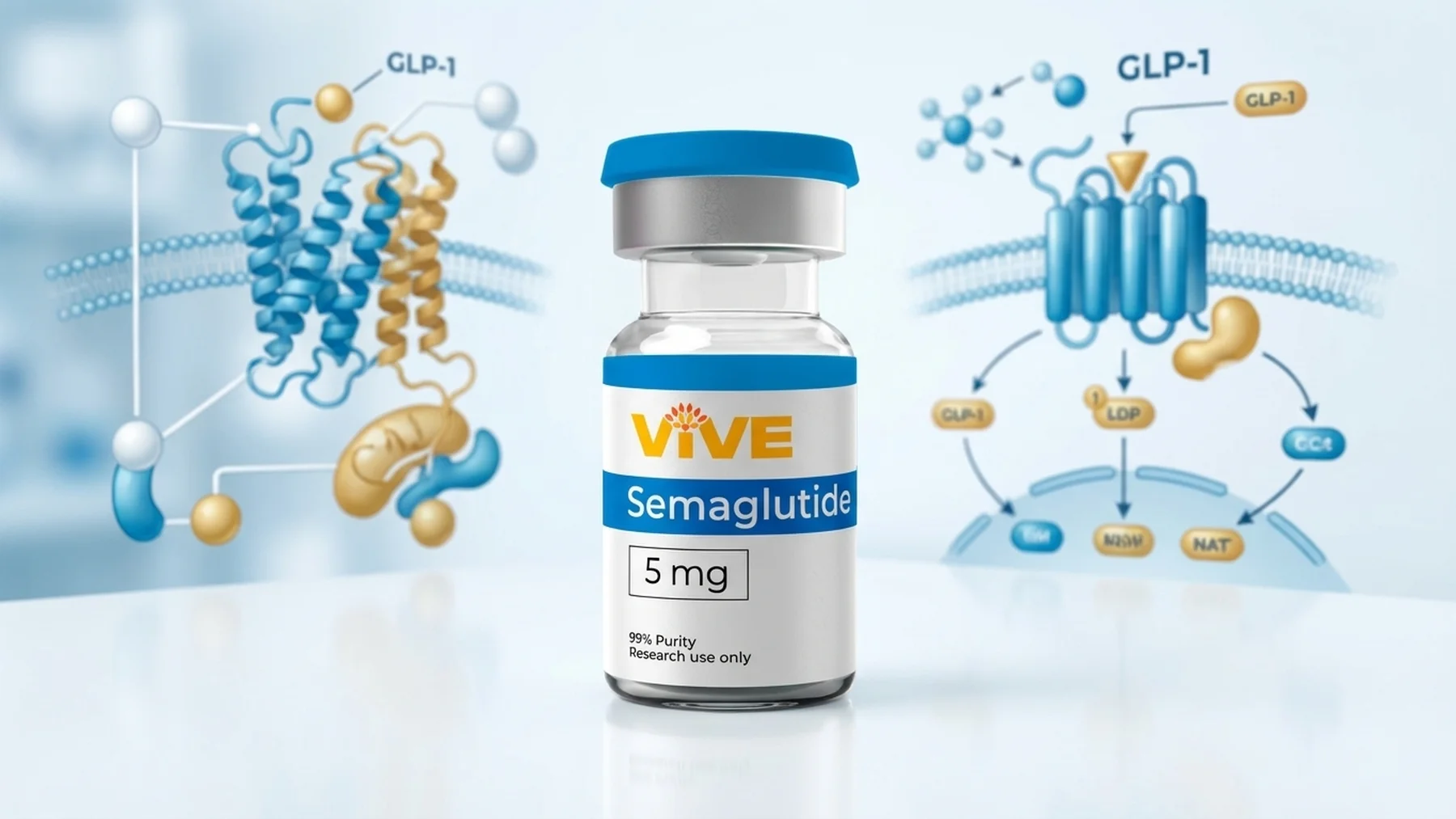
Semaglutide
Semaglutide is a GLP-1 receptor agonist with a modified C-18 fatty acid chain that extends its half-life to approximately one week through reversible albumin binding. The structural modification protects semaglutide from DPP-4 degradation while maintaining high GLP-1R binding affinity.
In research settings, semaglutide has been among the most studied GLP-1 compounds in terms of dose-response relationships, central versus peripheral receptor activation profiles, and long-term metabolic effects. A 2021 study by Wilding and colleagues published in The New England Journal of Medicine examined semaglutide’s effects on body weight in participants with obesity, establishing foundational data that researchers have used to build subsequent investigation into GLP-1-mediated weight regulation mechanisms.
Semaglutide is also notable as one of the first GLP-1 receptor agonists to receive regulatory review for both metabolic and cardiovascular indications. The SUSTAIN and STEP trial series generated extensive mechanistic data, including insights into GLP-1R-mediated anti-inflammatory signaling in vascular tissue.
VivePeptides supplies research-grade semaglutide at 99%+ purity, verified by independent HPLC and mass spectrometry analysis, with a batch-specific Certificate of Analysis provided with every order. Semaglutide from VivePeptides is intended for in-vitro laboratory research purposes only.
Tirzepatide
Tirzepatide is a dual receptor agonist that activates both GLP-1R and the glucose-dependent insulinotropic polypeptide receptor (GIP-R). This dual agonism represents a meaningful mechanistic difference from single GLP-1 receptor agonists. GIP receptor activation produces additive effects on insulin secretion and also acts on adipocyte receptors to modulate lipid metabolism, pathways that single-agonist compounds do not engage.
Research published by Jastreboff and colleagues in The New England Journal of Medicine (2022) examined tirzepatide’s effects in the context of obesity, and the findings generated significant interest in the GIP/GLP-1 synergy mechanisms involved. The dual receptor signaling produced through tirzepatide has made it a research compound of particular interest for studying the intersection of incretin biology and adipose tissue physiology.
Tirzepatide also offers researchers a comparison point for isolating GLP-1-specific versus GIP-specific receptor contributions to observed metabolic effects, a mechanistic question that has driven significant investigation into incretin receptor pharmacology.
VivePeptides offers research-grade tirzepatide verified to 99%+ purity with HPLC/MS confirmation and batch-specific COA documentation. For in-vitro laboratory research use only.
Liraglutide
Liraglutide was among the first long-acting GLP-1 receptor agonists to receive broad study. Its C-16 fatty acid attachment extends the half-life to approximately 13 hours. Mechanistically, liraglutide follows the same GLP-1R activation pathway as semaglutide, but its shorter half-life and different albumin-binding profile make it a distinct research compound for comparative pharmacokinetic studies.
Research on liraglutide published by Astrup and colleagues in The Lancet (2009) established foundational data on GLP-1-mediated appetite regulation and energy balance that shaped subsequent compound development. Liraglutide remains a widely used reference compound in GLP-1 research due to its extensive published safety and mechanistic data.
GLP-1 Compounds in Research: A Comparison Table
The following table summarizes the receptor binding profile, half-life, and primary research relevance of the major GLP-1 research compounds.
| Compound |
Receptor Targets |
Approximate Half-Life |
Primary Research Application |
| Semaglutide |
GLP-1R |
~7 days |
Metabolic regulation, obesity, cardiovascular signaling, appetite CNS studies |
| Liraglutide |
GLP-1R |
~13 hours |
GLP-1R pharmacokinetics, beta cell function, weight regulation |
| Exenatide |
GLP-1R |
~2.4 hours |
Short-acting GLP-1R reference, glucose-stimulated insulin studies |
| Tirzepatide |
GLP-1R + GIP-R |
~5 days |
Dual incretin agonism, adipose physiology, comparative incretin studies |
| Retatrutide |
GLP-1R + GIP-R + GCGR |
~6 days |
Triple agonism, energy expenditure, brown adipose activation |
GLP-1 vs. Dual and Triple Agonists in Research
The distinction between single, dual, and triple receptor agonists has become a key variable in current metabolic research. Single GLP-1 receptor agonists like semaglutide allow researchers to isolate GLP-1R-specific signaling. Dual agonists like tirzepatide introduce GIP receptor effects, enabling comparative studies of the two incretin pathways.
Triple agonists like retatrutide add glucagon receptor (GCGR) agonism to the GLP-1R and GIP-R targets. Glucagon receptor activation increases hepatic glucose output and thermogenesis, pathways that add complexity but also potential mechanistic leverage in obesity-related research. Understanding how GCGR activation interacts with GLP-1R and GIP-R signaling at the molecular level is an active area of investigation.
VivePeptides carries research-grade retatrutide for researchers exploring triple agonist mechanisms, available at 99%+ purity with batch-specific COA and HPLC/MS verification.
Research Applications and Key Findings
GLP-1 receptor agonists have moved from early incretin biology studies to a broad range of research applications. Below are selected research areas where these compounds have generated notable findings.
Metabolic and Obesity Research
The role of GLP-1 receptor agonists in obesity research has expanded substantially over the past decade. Studies have used these compounds to map the neural and hormonal circuits governing energy balance, food intake regulation, and adipose tissue metabolism.
Research published by Pi-Sunyer and colleagues in The New England Journal of Medicine (2015) examined weight outcomes in subjects with obesity using liraglutide, establishing early evidence for the CNS-mediated appetite suppression pathway now widely studied. Subsequent semaglutide trials under the STEP program (Wilding et al., The New England Journal of Medicine, 2021) extended this data significantly.
These findings have made GLP-1 compounds central tools in preclinical models of obesity, where researchers use them to study the metabolic consequences of sustained GLP-1R activation, the role of leptin and insulin co-signaling, and the contribution of gut-brain axis communication to energy homeostasis.
Cardiovascular Signaling Research
GLP-1 receptors are expressed in cardiac tissue, vascular endothelium, and macrophages. Research published by Marso and colleagues in The New England Journal of Medicine (2016) as part of the LEADER trial documented cardiovascular event data in semaglutide-related compound studies that prompted further mechanistic investigation into GLP-1R-mediated cardioprotective signaling.
Laboratory-based research has since examined how GLP-1R activation influences endothelial nitric oxide production, macrophage inflammation, and cardiomyocyte metabolism. These are active research areas, particularly for understanding the interaction between metabolic and cardiovascular disease pathways.
Neuroprotection and CNS Biology
GLP-1 receptors in the brain are expressed not only in appetite-regulating hypothalamic circuits but also in regions associated with neuroinflammation, dopamine signaling, and neuroprotection. Research published by Holst and colleagues in the Journal of Clinical Endocrinology and Metabolism has documented GLP-1R expression patterns in human brain tissue.
Preclinical studies using GLP-1 receptor agonists as research compounds have investigated whether GLP-1R activation can modulate neuroinflammatory pathways, dopaminergic circuits, and synapse protection. These are emerging research areas distinct from the metabolic pathway work that GLP-1 compounds are more commonly associated with.
Blood Sugar Level Regulation Mechanisms
Understanding how blood sugar levels are regulated through incretin signaling remains a core research application. The glucose-dependent nature of GLP-1R-mediated insulin secretion makes these compounds valuable tools for studying the kinetics of beta cell response under varying glycemic conditions.
Research using semaglutide and tirzepatide in in-vitro and animal models has mapped the relationship between GLP-1R signaling intensity, cAMP production, and the calcium flux events that drive insulin granule exocytosis. These mechanistic studies help build understanding of type 2 diabetes-related beta cell dysfunction at the cellular level.
Frequently Asked Questions About GLP-1 Receptor Agonist Research
What is the primary mechanism of action for GLP-1 receptor agonists?
GLP-1 receptor agonists bind to and activate the GLP-1 receptor, a G protein-coupled receptor expressed in pancreatic beta cells, the gastrointestinal tract, and the central nervous system. Activation produces cAMP-dependent signaling that modulates insulin secretion, gastric emptying, and appetite regulation. The mechanism is glucose-dependent in pancreatic tissue, meaning GLP-1R activation potentiates insulin release only when blood glucose is elevated.
How is semaglutide different from liraglutide structurally?
Both are GLP-1 receptor agonists but differ in fatty acid chain length and albumin binding properties. Semaglutide carries a C-18 fatty diacid modification that produces tighter albumin binding and a longer half-life of approximately seven days compared to liraglutide’s 13 hours. This structural difference makes semaglutide a distinct research compound for studies examining duration-of-action effects on GLP-1R signaling.
What makes tirzepatide different from single GLP-1 receptor agonists?
Tirzepatide is a dual receptor agonist targeting both GLP-1R and GIP-R. GIP receptor activation produces additional effects on adipocyte metabolism and synergistic insulin secretion that single GLP-1 receptor agonists do not generate. This dual agonism profile makes tirzepatide a valuable research compound for studies comparing the individual contributions of each incretin pathway.
Are GLP-1 compounds the same as Ozempic or Wegovy?
Ozempic and Wegovy are brand names for prescription semaglutide formulations that have undergone FDA approved review for specific clinical indications. Research-grade GLP-1 compounds like those supplied by VivePeptides are distinct from these formulations. Research-grade semaglutide is produced for in-vitro laboratory research use only and is not equivalent to, nor a substitute for, any approved drug product.
What is the research significance of GLP-1 drugs versus dual agonists?
Single-target GLP-1 compounds allow researchers to isolate GLP-1 receptor-specific effects. Dual and triple agonists introduce additional receptor pathways, enabling comparison of GLP-1R versus GIP-R versus glucagon receptor contributions to observed outcomes. The choice between single, dual, and triple agonist research compounds depends on the specific signaling pathway under investigation.
Browse Research-Grade GLP-1 Compounds
VivePeptides supplies research-grade GLP-1 receptor agonists and related peptide compounds for in-vitro laboratory research. Every product ships with a batch-specific Certificate of Analysis, HPLC and mass spectrometry verification, and temperature-controlled packaging to protect peptide integrity in transit.
Current GLP-1 research compound inventory includes:
- Semaglutide: 99%+ purity, batch COA, HPLC/MS verified
- Tirzepatide: dual GLP-1R/GIP-R agonist, 99%+ purity, batch COA
- Retatrutide: triple agonist (GLP-1R/GIP-R/GCGR), 99%+ purity, batch COA
All VivePeptides research compounds are intended for in-vitro laboratory research purposes only and are not intended for human or veterinary use.
Published by the VivePeptides Research Team. All compounds referenced are research-grade materials supplied for in-vitro laboratory use only. Nothing in this article constitutes medical advice, clinical guidance, or a recommendation for human use.